Videos
Downloads

Xiidra Patient Brochure
This resource provides patients with a comprehensive overview of Xiidra

BlinkRx Quick Guide
(for Commercial Patients)
This resource is about the BlinkRx program for patients with commercial insurance

BlinkRx Quick Guide (for Government-Insured Patients)
This resource is about the BlinkRx program for patients with government insurance, including Medicare Part D

Xiidra Savings Card
Get patients started with the My Savings Program by having them register for a co-pay card or activate an existing card online

Xiidra Sample Tier Exception Letter
Fill out this form to request a payer to cover Xiidra at a lower tier

Xiidra Sample Appeals Letter
Utilize this resource if a patient's insurance company denies coverage

Xiidra Letter of Medical Necessity (New Patient Start)
Use this template to request prior authorization or document medical necessity for treatment with Xiidra

Xiidra Letter of Medical Necessity (Patient Continuation)
Use this template to document medical necessity and request continuation of coverage for treatment with Xiidra

BlinkRx Guide
Download these step-by-step instructions on filling a prescription through BlinkRx

BlinkRx Fax Form
This form is used to e-prescribe Xiidra through BlinkRx. For more details, see the BlinkRx Guide

BlinkRx EHR Flashcard
This flashcard provides information on how to find BlinkRx in your electronic health record (EHR)

Identifying DED in Your Practice
This publication discusses the different manifestations of DED and how underlying inflammatory conditions can be a precursor of the disease
Request XIIDRA

Get Xiidra Samples
Log in or register for Bausch + Lomb MySampleCloset® to request samples and receive additional information on Xiidra
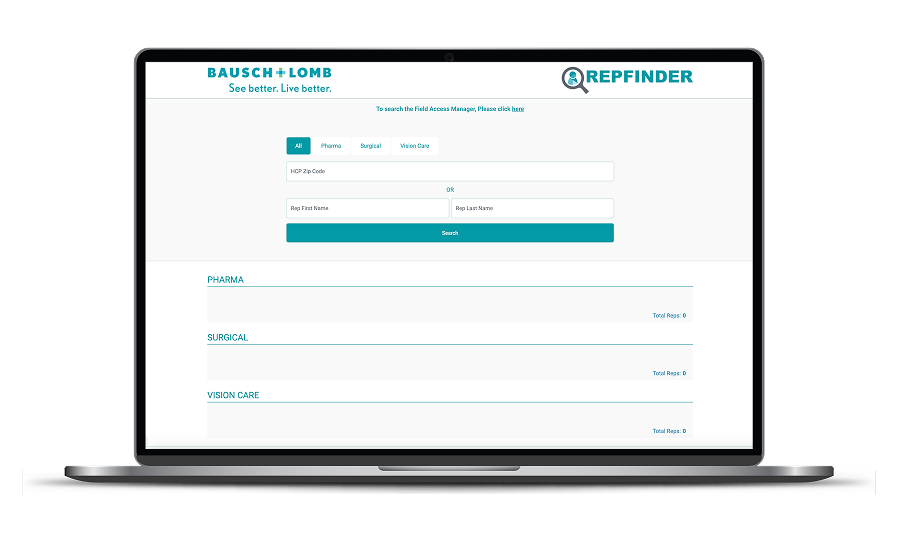
Request a Rep
Start by filling out the form with the Repfinder tool to search for a Field Access Manager in your area who could help you learn more about Xiidra.
FAQs
Xiidra is indicated for the treatment of the signs and symptoms of dry eye disease.1
- Xiidra is a prescription ophthalmic solution (eye drop) used in patients with dry eye disease to help treat both symptoms reported by patients and clinical signs observed by eye care professionals.1
- Its mechanism of action involves targeting the ICAM-1/LFA-1 interaction to help reduce inflammation associated with dry eye.1,2
Across 5 clinical studies, the most common AEs (reported in 5%–25% of patients) were instillation-site irritation, dysgeusia, and reduced visual acuity.1
Xiidra’s mechanism of action is thought to work by reducing inflammation on the surface of the eye that is associated with dry eye disease.1,2
- Xiidra targets the interaction between ICAM-1 and LFA-1.2
- By blocking the ICAM-1/LFA-1 interaction, Xiidra inhibits T-cell activation at multiple sites, reduces T-cell migration to the ocular surface, and reduces the release of proinflammatory cytokines.2
The exact mechanism of action of Xiidra is not known.
Xiidra is used once in the morning and once in the evening, approximately 12 hours apart.1
Contact lenses should be removed before using Xiidra and may be put back in 15 minutes after using the treatment.1
Xiidra is covered for ~77% of patients.* Find formulary coverage in your area by clicking here.
The cost for Xiidra will vary per patient. For patients who have limited or no prescription coverage, contact the Bausch + Lomb Patient Assistance Program at 855-770-0424 or visit www.patientassistance.bausch.com. For more on helping patients start and stay on Xiidra, and information on Xiidra's $0 Copay Program for commercially eligible patients or the Insider Support Program, please call the Xiidra Access Program at 1-877-494-4372.
BlinkRx assists with prior authorizations, cost, and customer support, offering a seamless experience for your patients. Find a helpful guide and additional downloadable resources about BlinkRx here.
There are a variety of ways to receive samples of Xiidra. You can visit and log in to Bausch + Lomb MySampleCloset®, call 1-800-746-1584, or use the “Request a Rep” button located above the main menu to connect with a representative.
Signs of dry eye inflammation can include conjunctival hyperemia, corneal and conjunctival staining, and tear hyperosmolarity.3 Symptoms of dry eye inflammation can include conjunctival redness and ocular pain, among others.2
XIIDRA FORMULARY
COVERAGE TOOL
Use this tool to help find coverage for Xiidra
in your area
XIIDRA WAS WELL
TOLERATED
Help patients get started by discussing
AEs and the safety profile of Xiidra1,2
AE, adverse event; ICAM-1, intercellular adhesion molecule 1; LFA-1, lymphocyte function-associated antigen 1.
The exact mechanism of action of Xiidra in dry eye disease is not known.
Xiidra significantly reduced symptoms of eye dryness at 2 weeks in 2 of 3 phase 3 studies, with improvements observed at 6 and 12 weeks in all 3 studies.1
*Based on MMIT data from January 2026.
References
- Xiidra. Prescribing Information. Bausch & Lomb Inc.
- Donnenfeld ED, Karpecki PM, Majmudar PA, et al. Safety of lifitegrast ophthalmic solution 5.0% in patients with dry eye disease: a 1-year, multicenter, randomized, placebo-controlled study. Cornea. 2016;35(6):741-748. doi:10.1097/ICO.0000000000000803
- Rao SK, Mohan R, Gokhale N, Matalia H, Mehta P. Inflammation and dry eye disease—where are we? Int J Ophthalmol. 2022;15(5):820-827. doi:10.18240/ijo.2022.05.20
Indication
Xiidra® (lifitegrast ophthalmic solution) 5% is indicated for the treatment of signs and symptoms of dry eye disease (DED).
Important Safety Information
- Xiidra is contraindicated in patients with known hypersensitivity to lifitegrast or to any of the other ingredients.
- In clinical trials, the most common adverse reactions reported in 5-25% of patients were instillation site irritation, dysgeusia and reduced