INFLAMMATION IS AN UNDERLYING CAUSE OF DRY EYE4
Stress to the ocular surface can cause an overexpression of inflammatory mediators, like ICAM-1. ICAM-1 binds to LFA-1, a cell surface protein found on T cells5,6
This interaction can lead to activation of T cells at multiple sites, migration of T cells to the ocular surface, and release of proinflammatory cytokines, triggering a vicious cycle of inflammation, which can lead to chronic tissue damage3,5,6
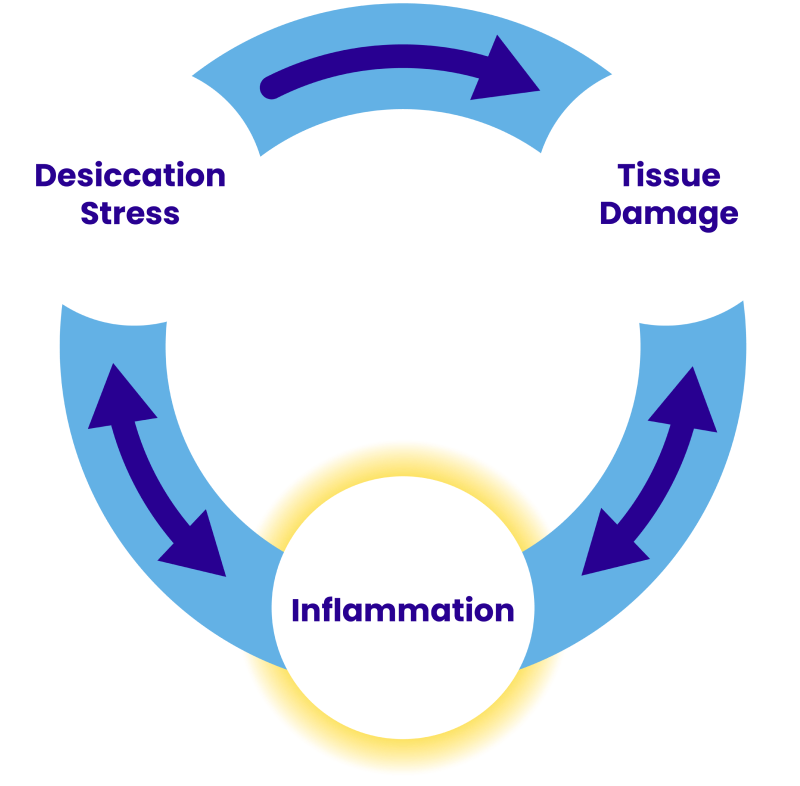
Up to 65% of patients with dry eye disease may have clinically significant ocular surface inflammation7
COMMON SYMPTOMS2,8
Patients living with dry eye disease may experience the following:
 Redness
Redness Burning
Burning Irritation
Irritation Ocular pain
Ocular pain Visual
Visual
fluctuation Foreign body sensation
Foreign body sensation
Symptoms such as ocular discomfort and signs such as conjunctival hyperemia, corneal and conjunctival staining, tear hyperosmolarity, and a positive MMP-9 test may indicate the presence of ocular surface inflammation3,9
Contributing Factors9
- Hormone imbalances (eg, menopausal age group)
- Prolonged contact lens use
- Underlying inflammatory conditions
- Autoimmune conditions (eg, Sjögren’s syndrome, rheumatoid arthritis)
- History of diabetes
- Ophthalmic surgery
Symptoms and risk factors of dry eye disease are not limited to those listed above.
Identifying Inflammation
The following scenarios represent real, ECP-provided case studies of patients who are experiencing dry eye with inflammation.
Clinical findings
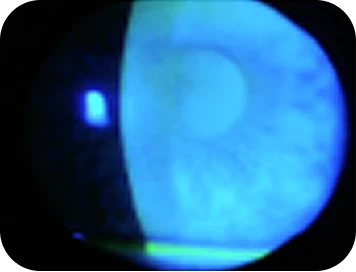
-
- Fluorescein staining: Moderate punctate epithelial erosions on cornea
- Corneal topography: Altered corneal curvature
Slit-lamp examination
-
- Mild to moderate conjunctival hyperemia OS
- Mild to moderate conjunctival staining
- Reduced TBUT
- No significant MGD
Background and medical history
- Wears disposable CLs
- Recently reduced CL wear time
- Mild seasonal allergies
Current symptoms and treatments
- Persistent ocular discomfort and EOD dryness
- Foreign body sensation
Current symptoms and treatments
- Intermittent blurred vision and visual disturbance
- Regular and preservative-free AT
Potential treatment goals
Reduced EOD eye
dryness and discomfort
Reduced dry
eye symptoms
Improved comfortable
CL wear time
CLs, contact lenses; MGD, meibomian gland dysfunction; OS, left eye; TBUT, tear breakup time.
Clinical findings
-
- Refraction: Soft 20/20 OU in phoropter
- Corneal staining: Mild/moderate
- TBUT: 2-3 seconds
Slit-lamp examination
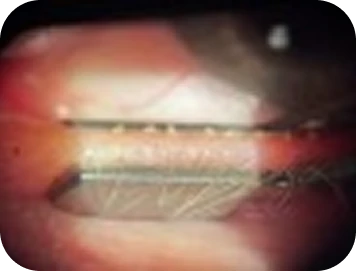
-
- Lashes: clear OU
- Lids: MGD – turbid gland expression OU
- Mild conjunctival hyperemia
- Punctate epithelial erosions on cornea
Background and medical history
- Postmenopausal
- Wears glasses
- Hypercholesterolemia, use of simvastatin
Current symptoms and treatments
- Mild redness OU
- Blurry vision at distance
Current symptoms and treatments
- Eye fatigue, burning, and watering
- Occasional use of ATs
Potential treatment goals
Improve fluctuating
vision and burning
Reduce corneal staining,
dryness, and foreign body
sensation
Increase ocular comfort
with prolonged reading
and screen use
MGD, meibomian gland dysfunction; OU, both eyes; TBUT, tear breakup time.
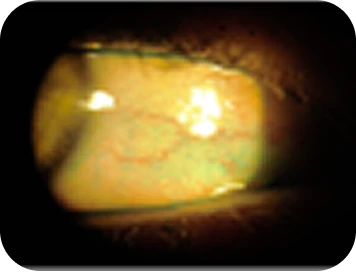
Clinical findings
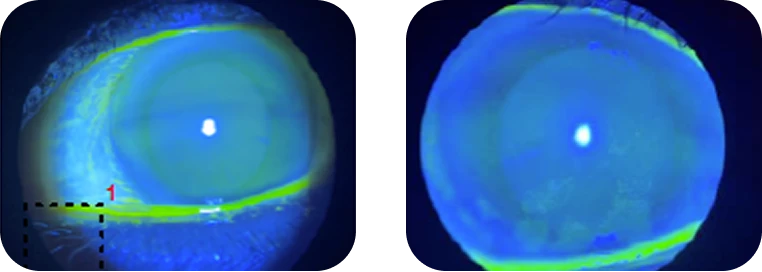
- Refractive errors; presbyopia and mild myopia
- SPEED score: 8
- Osmolarity: 310/325 mOsm
- InflammaDry: pos/pos
- Meibography: grade 2+ with inspissated discharge
- NaFl staining: 1+/3+ centrally
Background and medical history
- Scheduled for cataract surgery, multifocal IOLs
- Wears reading glasses for near-vision activities
Current symptoms and treatments
- History of refractive errors
- Pre-op assessment revealed DED
Current symptoms and treatments
- Mild eye dryness in the evening; frequent tired eye or vision fluctuation
- Difficulty adjusting to different light levels, driving, watching television
- Uses ATs regularly
Potential treatment goals
Improvement in ocular
surface health
Reduced corneal
staining
Improved biometry
accuracy
DED, dry eye disease; NaFl, sodium fluorescein; SPEED, Standardized Patient Evaluation of Eye Dryness.
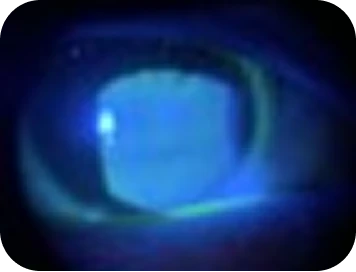
Clinical findings
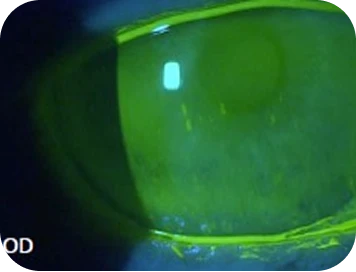
Slit-lamp examination
- Cornea staining: Grade 1+ in interpalpebral and central zones
- Conjunctival injection: Grade 2+
- TBUT: Reduced (8s): tear film instability
Slit-lamp examination
- SPEED score: 6
- Meibomian gland inspissation without scurf: Trace to 1+
- Tear osmolarity scores: 324 and 339 mOsm/L
Background and medical history
- No significant medical history of dry eye or related conditions other than mild and well-controlled hypertension
Current symptoms and treatments
- Redness and fluctuating vision that is worse toward the end of the workday
- Gritty and tired eyes
Current symptoms and treatments
- Complaints of burning and itchy eyes, especially after a day of computer use at work
- Has been using various ATs for the past few months with temporary and limited relief
Potential treatment goals
Symptom improvement
in 2 weeks
Sign improvement
Reduced AT use
SPEED, Standardized Patient Evaluation of Eye Dryness; TBUT, tear breakup time.
Clinical findings
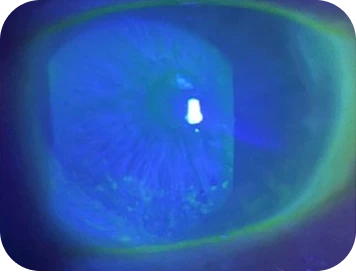
-
- Visual acuity: 20/40 OU
- InflammaDry test: positive for elevated MMP-9
Slit-lamp examination
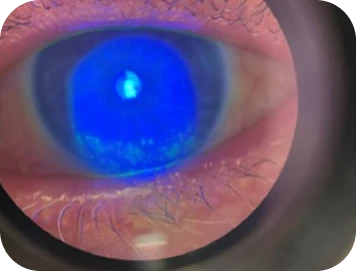
-
- Moderate conjunctival hyperemia
- Fluorescein staining: diffuse staining
Background and medical history
- Chronic eye discomfort for several years
- Occasional joint stiffness and fatigue, but no recent RA flares
Current symptoms and treatments
- Complaints of gritty sensation, redness, burning, and eye pain
- Visual fluctuations and chronic discomfort in both eyes
Current symptoms and treatments
- Uses ATs every 2-3 hours without relief
- Currently on disease-modifying therapy methotrexate and low-dose corticosteroids for RA
Potential treatment goals
Improvement in
symptoms of dry eye
Decrease in visual
fluctuation
Better tolerance for
prolonged visual task
OU, both eyes; QID, 4 times a day; RA, rheumatoid arthritis.
Patient portrayals. Images edited or rendered by AI.
How Xiidra Works
Xiidra addresses T cells differently to defend against dry eye inflammation6
ATs, artificial tears; EOD, end-of-day; ICAM-1, intercellular adhesion molecule 1; IOLs, intraocular lenses;
LFA-1, lymphocyte function-associated antigen 1; MMP-9, matrix metalloproteinase-9.
The exact mechanism of action of Xiidra in dry eye disease is not known.
References
- Craig JP, Nichols KK, Akpek EK, et al. TFOS DEWS II definition and classification report. Ocul Surf. 2017;15(3):276-283. doi:10.1016/j.jtos.2017.05.008
- American Academy of Ophthalmology. Dry Eye Syndrome Preferred Practice Pattern®. Accessed April 14, 2025. https://www.aaojournal.org/action/showPdf?pii=S0161-6420(18)32650-2
- Data on file. Bausch & Lomb Inc.
- Bron AJ, de Paiva CS, Chauhan SK, et al. TFOS DEWS II pathophysiology report. Ocul Surf. 2017;15(3):438-510. doi:10.1016/j.jtos.2017.05.011
- Pflugfelder SC, Stern M, Zhang S, Shojaei A. LFA-1/ICAM-1 interaction as a therapeutic target in dry eye disease. J Ocul Pharmacol Ther. 2017;33(1):5-12. doi:10.1089/jop.2016.0105
- Donnenfeld ED, Perry HD, Nattis AS, Rosenberg ED. Lifitegrast for the treatment of dry eye disease in adults. Expert Opin Pharmacother. 2017;18(14):1517-1524. doi:10.1080/14656566.2017.1372748
- Sambursky R. Presence or absence of ocular surface inflammation directs clinical and therapeutic management of dry eye. Clin Ophthalmol. 2016;10:2337-2343. doi:10.2147/OPTH.S121256
- Sullivan DA, Rocha EM, Aragona P, et al. TFOS DEWS II sex, gender, and hormones report. Ocul Surf. 2017;15(3):284-333. doi:10.1016/j.jtos.2017.04.001
- Market Scope. 2023 Dry Eye Products Market Report: Global Analysis for 2022 to 2028. Market Scope®, LLC; 2023. Accessed April 14, 2025. https://www.market-scope.com/pages/reports/411/2023-dry-eye-products-market-report-global-analysis-for-2022-to-2028-november-2023
Indication
Xiidra® (lifitegrast ophthalmic solution) 5% is indicated for the treatment of signs and symptoms of dry eye disease (DED).
Important Safety Information
- Xiidra is contraindicated in patients with known hypersensitivity to lifitegrast or to any of the other ingredients.
- In clinical trials, the most common adverse reactions reported in 5-25% of patients were instillation site irritation, dysgeusia and reduced





