ELIGIBLE COMMERCIAL Patients may pay as little as

with the Xiidra copay card or through BlinkRx†

Need to enroll your patient for a copay card?
Help your patients get started with the Xiidra My Saviings Program by having them register for a copay card or activate an existing card at xiidra.blsavingscard.com

Additional questions about cost and affordability?
For more on helping patients start and stay on Xiidra and information on the Xiidra My Saviings Program for eligible commercial patients, please call the copay program support line at 1-877-4XiiDRA (1-877-494-4372)
We’ve got you and your patients covered
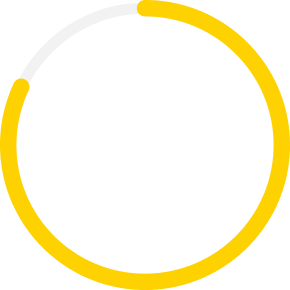
Xiidra is covered for 82% of dry eye
Commercial
- Xiidra is covered for 78% of commercial patients*
- 85% of covered commercial patients do not require step therapy*
Medicare Part D
- Xiidra is covered for 74% of Medicare patients*
- 94% of covered Medicare patients do not require prior authorization*
Uninsured
- Patients may pay no more than $225 with the Xiidra copay card or through BlinkRx‡
There are additional ways to help patients save

BlinkRx assists with prior authorizations and more, offering a seamless experience for your patients
Making Xiidra more accessible across coverage types
Prescribe through BlinkRx or provide your patients with the Xiidra copay card§ for use at retail pharmacies to receive the following benefits:
COMMERCIALLY INSURED
| Coverage Status | First Fill | Refills |
|---|---|---|
| COMMERCIALLY INSURED | ||
| Covered | $0 (30/60/90-day supply) |
$0 (30/60/90-day supply) |
| Not Covered | $0 (30-day supply) |
$0 if prior authorization is approved (30/60/90-day supply) If prior authorization is submitted and denied: $79 for 30-day supply If prior authorization is not submitted: $225 |
- PLEASE NOTE:
-
- Commercially insured patients with a high deductible or a high copay may pay ~$79 per 30-day supply and subsequent refills until deductible and/or out-of-pocket maximum is met.
- Copay card at Retail: Prior authorization must be submitted through PARx for patient to be eligible for Xiidra at $79.
- BlinkRx: Prior authorizations can be submitted through CoverMyMeds or PARx for patient to be eligible for Xiidra at $79.
GOVERNMENT INSURED
| GOVERNMENT INSURED | ||
| Coverage Status | First Fill | Refills |
|---|---|---|
| Coveredǁ | Plan copay (30/60/90-day supply) |
Plan copay (30/60/90-day supply) |
| Not Covered | If prior authorization is approved, plan copay | If prior authorization is approved, plan copay |
- EXCLUSIVELY THROUGH BLINKRx:
- Eligible patients may pay a reduced cash price of $225 for Xiidra. Patients voluntarily choose to pay cash and not submit the prescription claim to their Medicare plan for the remainder of the calendar year.
UNINSURED, NON-GOVERNMENT
| UNINSURED, NON-GOVERNMENT |
| Coverage Status | First Fill | Refills |
|---|---|---|
| Cash Pay | $225 (30-day supply) |
$225 (30-day supply) |
§Terms and conditions apply. See Eligibility Criteria and Terms and Conditions at xiidra.blsavingscard.com.
ǁCopay card is not applicable for covered government patients.

Is your patient experiencing financial hardship?
For patients who have limited or no prescription coverage, contact the Bausch + Lomb Patient Assistance Program at 855-770-0424 or visit www.patientassistance.bausch.com. Patients who are eligible may receive their medication for free
GET STARTED WITH
BLINKRx
Being proactive in the prior authorization process may help improve your patients’ success with Xiidra
Help Patients Start and Stay on Xiidra
Send patients home with the materials they need for a smooth onboarding process
*Based on MMIT data from January 2026.
†Offer not valid for patients whose prescriptions are reimbursed by any federal or state healthcare program, including Medicaid, Medicare, TRICARE, or any other federal or state healthcare program. Other restrictions apply. See complete Eligibility Criteria and Terms and Conditions at xiidra.blsavingscard.com.
‡For patients under the age of 65.
Indication
Xiidra® (lifitegrast ophthalmic solution) 5% is indicated for the treatment of signs and symptoms of dry eye disease (DED).
Important Safety Information
- Xiidra is contraindicated in patients with known hypersensitivity to lifitegrast or to any of the other ingredients.
- In clinical trials, the most common adverse reactions reported in 5-25% of patients were instillation site irritation, dysgeusia and reduced











